Signostics receives FDA clearance for world’s smallest ultrasound scanner
5 June 2009
Signostics has secured clearance from the US FDA for its palm-sized personal ultrasound product, the Signos. Signostics’ device makes it affordable for individual healthcare professionals to use ultrasound in their clinical practice. This breakthrough product is the first ultrasound platform technology that clinicians can place in their pocket or wear around their neck like a stethoscope.
Just as handheld devices transformed the business world, Signostics anticipates that the Signos will change the way healthcare professionals practice medicine.
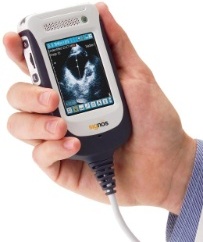 Retailing at a fraction of the cost of larger machines, the Signos is
the world’s smallest and lightest ultrasound product. With a weight of
approximately half a pound, its portability and low price point appeals
to a large range of medical markets, including emergency medicine,
primary care, remote healthcare, critical care, pediatrics, musculo-skeletal,
palliative care and sports medicine.
Retailing at a fraction of the cost of larger machines, the Signos is
the world’s smallest and lightest ultrasound product. With a weight of
approximately half a pound, its portability and low price point appeals
to a large range of medical markets, including emergency medicine,
primary care, remote healthcare, critical care, pediatrics, musculo-skeletal,
palliative care and sports medicine.
“Securing FDA clearance is a huge milestone for our company,” said Signostics Chief Executive Officer Dr Neil Bartlett. “The global financial crisis over the last six months has put pressure on health costs around the world and increased the need for more affordable medical products that can deliver results rapidly and conveniently. The Signos meets these requirements and physician interest in the product at industry events and among industry leaders has been remarkable. We are very optimistic about the sales outlook for the company.”
According to McGregor Tan Research in their 2005 Market Assessment, the global personal ultrasound market is estimated to be worth around $3 billion, with the US market worth about $1 billion.
“Signostics overcame some difficult product design challenges in developing its innovative palm-sized, portable ultrasound product,” said Patrick O’Doherty, healthcare segment director, Analog Devices, Inc. (NYSE: ADI), a global leader in high-performance semiconductors for signal-processing applications and longtime collaborator with the medical imaging industry.
“Analog Devices worked closely with Signostics to provide key signal processing technologies for the data conversion, signal conditioning and sensing necessary to achieve their design. We congratulate Signostics on receiving FDA clearance to market the revolutionary Signos personal ultrasound product.”
The Signos delivers high-resolution images anywhere, at any time for general medical applications, including: abdominal assessments such as bladder, abdominal aortic aneurysm screening, and trauma assessment; musculoskeletal; and basic obstetrics, such as pregnancy viability and fetal positioning.
Signostics has received funding from angel investors, Australian government grant providers and Playford Capital. The company will shortly close a round of funding at almost $4 million, having raised a total of $12 million to date.
The brainchild of physician-engineer Dr Neil Bartlett, Signostics was established in Adelaide, Australia in January 2005. Signostics employs a cutting-edge development team including physicians, sonographers as well as biomedical, electronic and software engineers. Signostics’ global sales and marketing headquarters is located in the United States in Silicon Valley, California.
Bookmark this page